Featured Publication:
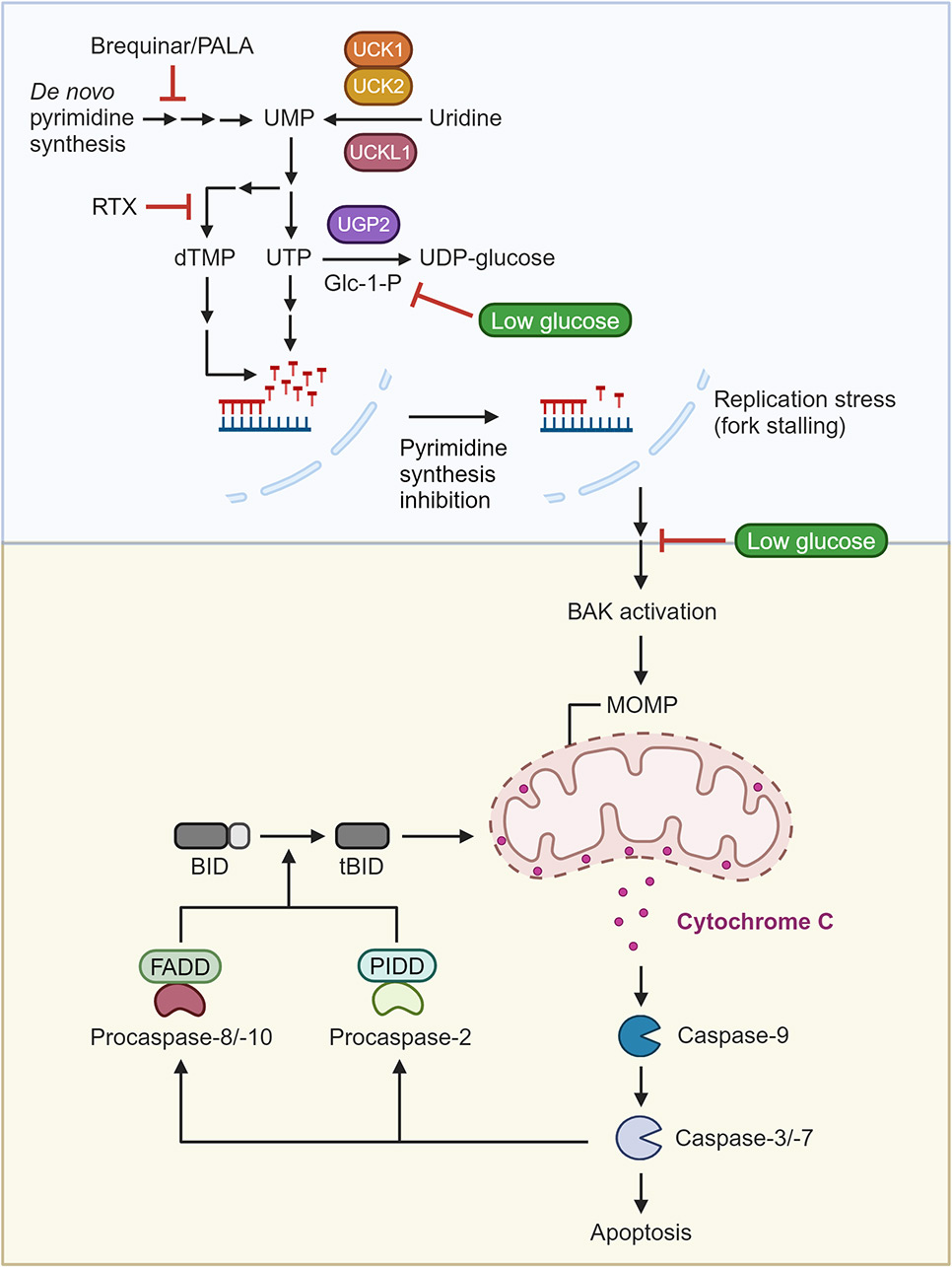
Glucose limitation protects cancer cells from apoptosis induced by pyrimidine restriction and replication inhibition
Nam M, Xia W, Mir AM, Jerrett A, Spinelli JB, Huang TT, Possemato R. Nature Metabolism. 2024 Nov 26. doi: 10.1038/s42255-024-01166-w.
Cancer cells often experience nutrient-limiting conditions because of their robust proliferation and inadequate tumour vasculature, which results in metabolic adaptation to sustain proliferation. Most cancer cells rapidly consume glucose, which is severely reduced in the nutrient-scarce tumour microenvironment. In CRISPR-based genetic screens to identify metabolic pathways influenced by glucose restriction, we find that tumour-relevant glucose concentrations (low glucose) protect cancer cells from inhibition of de novo pyrimidine biosynthesis, a pathway that is frequently targeted by chemotherapy. We identify two mechanisms to explain this result, which is observed broadly across cancer types. First, low glucose limits uridine-5-diphosphate-glucose synthesis, preserving pyrimidine nucleotide availability and thereby prolonging the time to replication fork stalling. Second, low glucose directly modulates apoptosis downstream of replication fork stalling by suppressing BAK activation and subsequent cytochrome c release, key events that activate caspase-9-dependent mitochondrial apoptosis. These results indicate that the low glucose levels frequently observed in tumours may limit the efficacy of specific chemotherapeutic agents, highlighting the importance of considering the effects of the tumour nutrient environment on cancer therapy.
Key Publications

Allosteric regulation of CAD modulates de novo pyrimidine synthesis during the cell cycle
Shin J, Mir H, Khurram MA, Fujihara KM, Dynlacht BD, Cardozo TJ, Possemato R. Nat Metab. 2023 Feb 6. doi: 10.1038/s42255-023-00735-9.
Metabolism is a fundamental cellular process that is coordinated with cell cycle progression. Despite this association, a mechanistic understanding of cell cycle phase-dependent metabolic pathway regulation remains elusive. Here we report the mechanism by which human de novo pyrimidine biosynthesis is allosterically regulated during the cell cycle. Combining traditional synchronization methods and metabolomics, we characterize metabolites by their accumulation pattern during cell cycle phases and identify cell cycle phase-dependent regulation of carbamoyl-phosphate synthetase 2, aspartate transcarbamylase and dihydroorotase (CAD), the first, rate-limiting enzyme in de novo pyrimidine biosynthesis. Through systematic mutational scanning and structural modelling, we find allostery as a major regulatory mechanism that controls the activity change of CAD during the cell cycle. Specifically, we report evidence of two Animalia-specific loops in the CAD allosteric domain that involve sensing and binding of uridine 5'-triphosphate, a CAD allosteric inhibitor. Based on homology with a mitochondrial carbamoyl-phosphate synthetase homologue, we identify a critical role for a signal transmission loop in regulating the formation of a substrate channel, thereby controlling CAD activity.
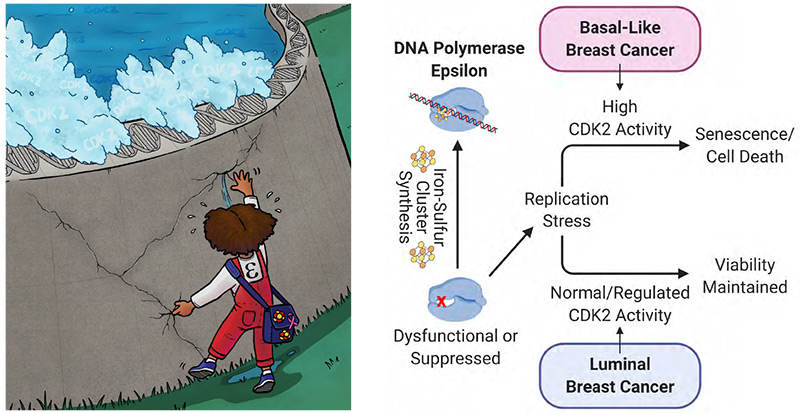
Hyperactive CDK2 Activity in Basal-like Breast Cancer Imposes a Genome Integrity Liability that can be Exploited by Targeting DNA Polymerase Epsilon
Sviderskiy VO, Blumenberg L, Gorodetsky E, Karakousi TR, Hirsh N, Alvarez SW, Terzi EM, Kaparos E, Whiten GC, Ssebyala S, Tonzi P, Mir H, Neel BG, Huang TT, Adams S, Ruggles KV, and Possemato R. Molecular Cell, 2020 Nov 19; 80:1-17.
- Basal-like breast cancer (BLBC) cells are especially sensitive to suppression of iron-sulfur cluster (ISC) biosynthesis, compared to other breast cancer sub-types or non-transformed mammary epithelial cells.
- The catalytic subunit of DNA polymerase epsilon (POLE1) requires ISC cofactor binding to support replication and cell viability. BLBC is selectively susceptible to partial suppression of POLE1, but not the other replicative polymerases, indicating that POLE1 is the ISC protein that underlies the sensitivity of this breast cancer subtype to suppression of ISC synthesis.
- Suppression of POLE1 results in DNA damage, expression of inflammatory cytokines, and a senescence-like state or cell death, selectively in BLBC cells.
- Non-BLBC cells and non-transformed mammary epithelial cells depend on properly regulated activity of the cyclin dependent kinase CDK2 to maintain viability when POLE1 or ISC synthesis is suppressed.
- Human BLBC tumors have increased phosphorylation of CDK2 target genes compared to other breast cancer sub-types, consistent with genetic alterations that promote CDK2 activity in BLBC.
- Hyperactive CDK2 may underlie the sensitivity of BLBC cells to ISC or POLE1 suppression, and may represent a liability for BLBC cells that can be targeted for anti-cancer therapy.
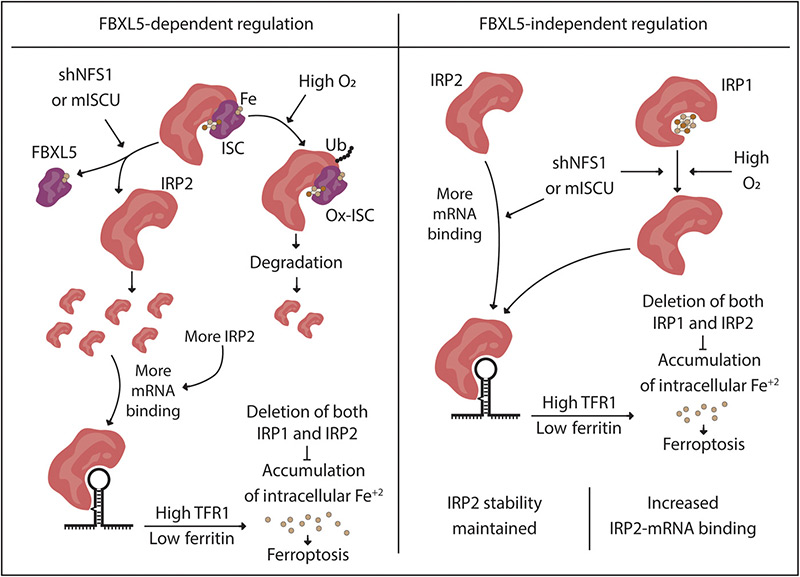
Iron-sulfur cluster deficiency can be sensed by IRP2 and regulates iron homeostasis and sensitivity to ferroptosis independent of IRP1 and FBXL5.
Terzi EM, Sviderskiy VO, Alvarez SW, Whiten GC, Possemato R.. Science Advances. 2021 May 26;7(22):eabg4302.
- Inhibiting Iron-Sulfur cluster (ISC) synthesis triggers sensitivity to ferroptosis in cancer cells that upregulate the transferrin receptor We show that most breast and lung cancer cell lines initiate an iron-starvation response upon ISC synthesis inhibition. Those that trigger this response exhibit increased ferroptosis sensitivity. Inhibition of upregulation of the transferrin receptor is sufficient to block this increased ferroptosis sensitivity.
- IRP1 is dispensable for altered iron metabolism upon ISC inhibition. Prior work indicated that IRP1 would be responsible for modulating iron metabolism upon ISC synthesis inhibition. We show in IRP1 null cells that the iron-starvation response upon ISC synthesis inhibition is still intact, and cell retain ferroptosis sensitivity.
- IRP2 stability is upon ISC synthesis inhibition is dependent upon environmental oxygen level. Prior work indicated that IRP2 stability is increased upon ISC synthesis inhibition and hypothesized that this occurs via an IRP1-dependent mechanism. Here, we show that IRP2 stability upon ISC synthesis inhibition is dependent upon environmental oxygen independent of IRP1. We hypothesize that a ubiquitin ligase known to regulate IRP2, FBXL5, is more active at elevated oxygen concentrations as delineated by a recent report (Wang et al, Molecular Cell, 2020).
- IRP2 can activate the iron starvation response and sensitize cells to ferroptosis independent of changes in protein level. IRP2 binding to mRNAs involved in iron metabolism is stimulated by ISC synthesis inhibition, independent of FBXL5 or changes in IRP2 stability at tissue-relevant oxygen concentrations.
- Cells lacking both IRP1 and IRP2 are resistant to ferroptosis. Deletion of both IRP1 and IRP2 renders cells dependent upon iron supplementation or forced expression of the transferrin receptor. Cell lacking both IRP1 and IRP2 are resistant to ferroptosis, even when ISC synthesis is inhibited.
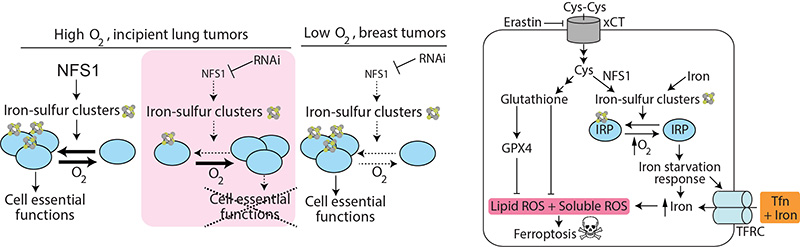
NFS1 Undergoes Positive Selection in Lung Tumors and Protects Cells From Elevated Oxygen Levels and Ferroptosis
Alvarez SW*, Sviderskiy VO*, Terzi EM, Papagiannakopoulos T, Moreira AL, Adams S, Sabatini DM, Birsoy K, and Possemato R. Nature. 2017 Nov 30.
- Oxygen level is a key factor in understanding the response to metabolic perturbation. We describe how cancer cells in culture are more sensitive to suppression of enzymes utilizing oxygen as a substrate compared to these same cells in a tumor. These data suggest that growing cells in culture at tissue level oxygen would be one simple and important way to mimic in vivo conditions.
- Lung tumors select for a metabolic pathway that protects against damage from a nutrient in the tumor microenvironment. We demonstrate that NFS1, a critical enzyme in the biosynthesis of iron-sulfur cluster cofactors, is upregulated in lung adenocarcinoma and when cells are placed in high oxygen environments. Iron-sulfur clusters are present in numerous cell-essential proteins and are sensitive to oxidative damage.
- Physiologic changes in oxygen level affect iron-sulfur cluster proteins more than exogenous oxidants do. We describe how iron-sulfur cluster containing proteins lose activity in high oxygen, but not upon moderate stress with hydrogen peroxides. Similarly, suppressing iron-sulfur cluster cofactor production blocks proliferation, which can be rescued by low oxygen culture, but not by anti-oxidants.
- Suppressing iron-sulfur cluster biosynthesis activates the iron-starvation response and increases sensitivity to oxidative cell death, also known as ferroptosis. We show that the classic iron-starvation response (increase in iron uptake and intracellular iron release) is dramatically upregulated upon iron-sulfur cluster biosynthesis suppression. Treatment with exogenous oxidants results in a type of cell death called ferroptosis, likely because the increase in free intracellular iron potentiates oxidative damage by producing superoxide radicals. These results lead us to the hypothesis that activating the iron-starvation response can trick cancer cells into increasing iron uptake, leaving them vulnerable to death by ferroptosis.
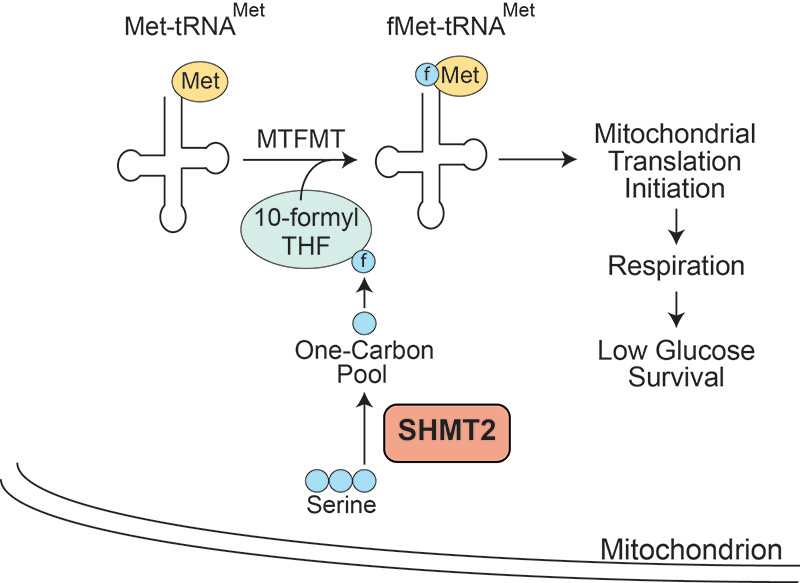
Serine Catabolism by SHMT2 Is Required for Proper Mitochondrial Translation Initiation and Maintenance of Formylmethionyl-tRNAs
Minton DR, Nam M, McLaughlin DJ, Shin J, Bayraktar EC, Alvarez SW, Sviderskiy VO, Papagiannakopoulos T, Sabatini DM, Birsoy K, Possemato R. Serine Catabolism by SHMT2 Is Required for Proper Mitochondrial Translation Initiation and Maintenance of Formylmethionyl-tRNAs. Mol Cell. 2018 Feb 15;69(4):610-621.
- A mitochondrial pathway that catabolizes the amino acid serine is required to support mitochondrial function. Mitochondrial one-carbon metabolism harvests carbon units from serine for use in several metabolic reactions requiring one carbon donation, particularly in the de novo synthesis of nucleotide bases. We show that complete loss of this pathway also impairs mitochondrial oxidative phosphorylation because of a loss of mitochondrially encoded proteins. A parallel pathway of one carbon metabolism in the cytoplasm can largely make up for loss of the mitochondrial pathway by supporting nucleotide metabolism, but cannot provide sufficient one carbon units to the mitochondria to maintain its function. These findings indicate that cells maintain two parallel pathways in the mitochondrial and cytoplasm to support this critical mitochondrial function.
- Carbon units harvested from serine are used to modify mitochondrial tRNAs, supporting translation in the mitochondria. The mitochondrial genome encodes 13 proteins involved in oxidative phosphorylation, which are transcribed and translated in the mitochondria. Prokaryotes and mitochondrial ribosomes, but not eukaryotic cytoplasmic ribosomes, prefer to start translation with a modified initiator tRNA in which the methionine is formylated. We show that this formyl group originates from the amino acid serine via mitochondrial one carbon metabolism. Complete blockade of mitochondrial one carbon metabolism impairs production of formyl-methionine tRNAs, which results in the loss of translation of several mitochondrial proteins.
Metabolic determinants of cancer cell sensitivity to glucose limitation and biguanides
Birsoy and Possemato et al, Nature, 2014 Apr 3.
- Continuous flow cell culture can be used to maintain cells in limiting nutrient conditions for extended periods, permitting genetic screening in these conditions. We describe the development of an apparatus that we call the Nutrostat, in which cells are maintained in 1 mM glucose for weeks. These conditions permit exponential growth approaching nutrient replete levels and high-throughput screening.
-
Cell-barcoding can be used to perform competition assays between cell lines in nutrient limiting conditions. We develop a technique to compare the sensitivity of cell lines to changes in the nutrient levels of the environment. Dozens of non-adherent cell lines are tagged by stable barcodes transduced lentivirally. We show that these cell lines can be grown together in large cultures to simultaneously measure the differential response of these lines to glucose limitation.
Cancer cells grown under low glucose conditions require robust oxidative phosphorylation. Using loss-of-function genetic screening in Nutrostats, we show that genes encoding core components of oxidative phosphorylation complexes are differentially required in low glucose culture. - Cancer cell lines sensitive to low glucose culture harbor mutations in mitochondrially encoded proteins or exhibit low expression of genes required for glucose utilization. Using the cell line barcoding technique described above, we show that cell lines with an inability to activate oxidative phosphorylation in response to glucose limitation exhibit exquisite sensitivity to low glucose culture. These cell lines either harbor loss-of-function mutations in mitochondrially encoded proteins, which encode core subunits of oxidative phosphorylation, or express low levels of glycolytic genes and glucose transporters.
- Cancer cell lines sensitive to low glucose culture are sensitive to biguanide treatment. Biguanides such as metformin and phenformin can inhibit mitochondrial oxidative phosphorylation Complex I. Indeed, we show that the effects of phenformin on oxygen consumption, and its toxicity on cells in culture and on xenograft tumors can be rescued by the expression of a yeast protein, Ndi1, that can bypass Complex I. Cell lines harboring mutations in mitochondrial proteins or expressing low levels of glycolytic genes and glucose transporters are particularly sensitive to phenformin treatment. These data suggest that in tumors that harbor such mutations in the mitochondrial genome, biguanide treatment may be efficacious.
Functional genomics reveal that the serine synthesis pathway is essential in breast cancer
Possemato et al, Nature, 2011 Aug 18.
- Serine synthesis is upregulated in basal-like breast cancer. We show that basal-like breast tumors exhibits high expression of the enzymes of serine biosynthesis, especially phosphoglycerate dehydrogenase (PHGDH). Cell lines derived from these tumors convert metabolites of glucose to serine, and can also import serine from the environment. Serine is consumed for several important downstream biosynthetic pathways including nucleotide metabolism, membrane lipid synthesis, and synthesis of the amino acids glycine and cysteine. PHGDH is one of 50 genes whose expression is used in a common classifier of breast cancer into subtypes, the PAM50, because it is highly specific for basal-like disease. Subsequent work has shown that expression of serine biosynthetic enzymes is required in a variety of tumor types to support downstream biosynthetic pathways.
- Activation of serine synthesis drives flux into branching metabolic pathways. We demonstrate that by increasing serine biosynthesis, basal-like breast cancer cells also increase flux of glutamine into the TCA cycle, as transamination by PSAT1 also results in the conversion of glutamate to alpha-ketoglutarate. In fact, in serine synthesis high cell lines, the PSAT1 reaction is responsible for about half of the flux from glutamine into the TCA cycle.
- Serine synthesis is silenced in a subset of breast tumors, particularly luminal subtypes, and cancer cell lines exhibiting silencing of these genes are dependent on exogenous serine. We show that some cell lines with low expression of PHGDH, PSAT1, or PSPH, are serine auxotrophs, which are dependent upon serine uptake.
Selected Publication:
View ArticleTerzi EM, Sviderskiy VO, Alvarez SW, Whiten GC, Possemato R. Iron-sulfur cluster deficiency can be sensed by IRP2 and regulates iron homeostasis and sensitivity to ferroptosis independent of IRP1 and FBXL5. Science Advances. 2021 May 26;7(22):eabg4302.
View ArticleMinton DR NM, McLaughlin DJ, Shin J, Bayraktar EC, Alvarez SW, Sviderskiy VO, Papagiannakopoulos T, Sabatini DM, Birsoy K, Possemato R. Serine Catabolism by SHMT2 is Required for Proper Mitochondrial Translation Initiation and Maintenance of Formylmethionyl tRNAs. Molecular cell.
View Article Download PDFAlvarez SW and Possemato R. Leveraging the iron-starvation response to promote ferroptosis, Oncotarget, 2018, Vol. 9, (No. 13), pp: 10830-10831.
View ArticleVaeth M, Maus M, Klein-Hessling S, Freinkman E, Yang J, Eckstein M, Cameron S, Turvey SE, Sefling E, Berberich-Siebelt F, Possemato R, Feske S. Store-operated Ca2+ entry, calcineurin and NFAT control clonal expansion of T cells through metabolic reprogramming, Immunity, 2017 Oct 17;47(4):664-679.e6.
View Article Download PDFSayin V, LeBoeuf S, Singh S, Davidson S, Biancur D, Guzelhan B, Alvarez SW, Wu WL, Karakousi TR, Zavitsanou AM, Ubriaco J, Muir A, Karagiannis D, Morris PJ, Thomas CJ, Possemato R, Vander Heiden MG, Papagiannakopoulos T. Activation of the NRF2 antioxidant program causes defects in central carbon metabolism, eLife, 2017;6:e28083.
View Article Download PDFPacold ME, Brimacombe KR, Chan SH, Rohde JM, Lewis CA, Swier LJ, Possemato R, Chen WW, Sullivan LB, Fiske BP, Cho S, Freinkman E, Birsoy K, Abu-Remaileh M, Shaul YD, Liu CM, Zhou M, Koh MJ, Chung H, Davidson SM, Luengo A, Wang AQ, Xu X, Yasgar A, Liu L, Rai G, Westover KD, Vander Heiden MG, Shen M, Gray NS, Boxer MB, Sabatini DM. A PHGDH inhibitor reveals coordination of serine synthesis and one-carbon unit fate. Nat Chem Biol. 2016 Jun;12(6):452-8.
View Article Download PDFTsun ZY, Possemato R. Amino Acid Management in Cancer. Semin Cell Dev Biol. 2015 Aug 12 pii: S1084-9521(15)00146-9.
View Article Download PDFStrohecker A, Joshi S, Possemato R, Abraham R, Sabatini DM, White E. Identification of 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase (PFKFB4) as a Novel Autophagy Regulator by High Content shRNA Screening. Oncogene. 2015 Mar 16.
View Article Download PDFKim D, Fiske BP, Birsoy K, Kami K, Possemato R, Chudnovsky Y, Pacold ME, Freinkman E, Chen WW, Shelton LM, Gui DY, Kwon M, Kang SW, Snuderl M, Vander Heiden MG, Sabatini DM. SHMT2 drives glioma cell survival in the tumor microenvironment but imposes a liability for glycine toxicity. Nature. 2015 Apr 8.
View Article Download PDFShaul YD, Freinkman E, Comb WC, Cantor JR, Tam WL, Thiru P, Kim D, Kanarek N, Pacold ME, Chen WW, Bierie B, Possemato R, Reinhardt F, Weinberg RA, Yaffe MB, Sabatini DM. Dihydropyrimidine accumulation is required for the epithelial-mesenchymal transition. Cell. 2014;158(5):1094-109.
View Article Download PDFBirsoy K*, Possemato R*, Lorbeer FK, Bayraktar EC, Thiru P, Yucel B, Wang T, Chen WW, Clish CB, Sabatini DM. Metabolic determinants of cancer cell sensitivity to glucose limitation and biguanides. Nature. 2014;508(7494):108-12.
View Article Download PDFGrabiner BC, Nardi V, Birsoy K, Possemato R, Shen K, Sinha S, Jordan A, Beck AH, Sabatini DM. A diverse array of cancer-associated MTOR mutations are hyperactivating and can predict rapamycin sensitivity. Cancer discovery. 2014;4(5):554-63.
View Article Download PDFBirsoy K, Wang T, Possemato R, Yilmaz OH, Koch CE, Chen WW, Hutchins AW, Gultekin Y, Peterson TR, Carette JE, Brummelkamp TR, Clish CB, Sabatini DM. MCT1-mediated transport of a toxic molecule is an effective strategy for targeting glycolytic tumors. Nature genetics. 2013;45(1):104-8.
View Article Download PDFNguyen CL*, Possemato R* Bauerlein EL, Xie A, Scully R, Hahn WC. Nek4 regulates entry into replicative senescence and the response to DNA damage in human fibroblasts. Molecular and cellular biology. 2012;32(19):3963-77.
View Article Download PDFBirsoy K, Sabatini DM, Possemato R. Untuning the tumor metabolic machine: Targeting cancer metabolism: a bedside lesson. Nature medicine. 2012;18(7):1022-3.
View Article Download PDFPossemato R, Marks KM, Shaul YD, Pacold ME, Kim D, Birsoy K, Sethumadhavan S, Woo HK, Jang HG, Jha AK, Chen WW, Barrett FG, Stransky N, Tsun ZY, Cowley GS, Barretina J, Kalaany NY, Hsu PP, Ottina K, Chan AM, Yuan B, Garraway LA, Root DE, Mino-Kenudson M, Brachtel EF, Driggers EM, Sabatini DM. Functional genomics reveal that the serine synthesis pathway is essential in breast cancer. Nature. 2011;476(7360):346-50.
View Article Download PDFMaida Y, Yasukawa M, Furuuchi M, Lassmann T, Possemato R, Okamoto N, Kasim V, Hayashizaki Y, Hahn WC, Masutomi K. An RNA-dependent RNA polymerase formed by TERT and the RMRP RNA. Nature. 2009;461(7261):230-5.
View Article Download PDFPossemato R, Timmons JC, Bauerlein EL, Wada N, Baldwin A, Masutomi K, Hahn WC. Suppression of hPOT1 in diploid human cells results in an hTERT-dependent alteration of telomere length dynamics. Molecular cancer research : MCR. 2008;6(10):1582-93.
View Article Download PDFMasutomi K, Possemato R, Wong JM, Currier JL, Tothova Z, Manola JB, Ganesan S, Lansdorp PM, Collins K, Hahn WC. The telomerase reverse transcriptase regulates chromatin state and DNA damage responses. Proceedings of the National Academy of Sciences of the United States of America. 2005;102(23):8222-7.
View Article Download PDFChen W*, Possemato R*, Campbell KT, Plattner CA, Pallas DC, Hahn WC. Identification of specific PP2A complexes involved in human cell transformation. Cancer cell. 2004;5(2):127-36.
View Article Download PDFPossemato R, Eggan K, Moeller BJ, Jaenisch R, Jackson-Grusby L. Flp recombinase regulated lacZ expression at the ROSA26 locus. Genesis. 2002;32(2):184-6.
View Article Download PDFJackson-Grusby L, Beard C, Possemato R, Tudor M, Fambrough D, Csankovszki G, Dausman J, Lee P, Wilson C, Lander E, Jaenisch R. Loss of genomic methylation causes p53-dependent apoptosis and epigenetic deregulation. Nature genetics. 2001;27(1):31-9.

